Interested in participating? Learn about a clinical study opportunity.

Phase 3 Clinical Study for Children and Adolescents Living With Angelman Syndrome (AS).

You are 4 to less than 18 years of age
Has confirmed deletion of the maternal copy of the UBE3A gene causing AS

Can walk independently or with an assistive device
If your child has been previously treated with an ASO or gene therapy, they may not be eligible to participate in this study. Please discuss any previous treatments with the study team to see if your child may be able to participate.
To help researchers learn more about the safety of GTX-102* and the effects it may have on:
* GTX-102 is an investigational antisense oligonucleotide (ASO) that is not currently approved by any health authority. However, it has been given to children and adolescents in a prior study.
For more information, please visit clinicaltrials.gov


GTX-102 or standard AS care‡
GTX-102
‡ Double-blind study treatment is assigned at random, with an equal chance of each option. The Double-blind Period will include a Sham Control group.


Assessments, testing, and study treatment (GTX-102) or Sham Control procedure (study procedure with no drug administration)
Discussion about changes in your child’s health, other medications/therapies

Physical exams

Neurological exams

Heart testing (electrocardiograms)

Blood, urine, and spinal fluid sample collection

Lumbar puncture (possible use of sedation)

AS functional assessments
Questionnaires for caregivers

Brainwave testing (EEG) and Video Assessments for some participants
Clinical Study for Individuals Living with Angelman Syndrome (AS)
Evaluating an investigational therapy called GTX-102 as a potential treatment.
To be eligible for study participation, individuals with AS will need to meet 1 set of the following requirements, depending on their age group:

1 to 3 years of age:
Confirmed diagnosis of deletion-type AS
Weight of at least 8 kg (17.6 lb.) at screening

4 to 17 years of age:
Confirmed diagnosis of paternal uniparental disomy (UPD) AS or imprinting center defect (ICD) AS

18 to 64 years of age:
Confirmed diagnosis of any AS genotype
If the individual has been previously treated with an ASO or gene therapy, they will not be eligible to participate in this study. Please discuss any previous treatments with the study team to see if your loved one may be able to participate. There are some additional requirements, which the study team will explain to you.
GTX-102 has been evaluated in a prior clinical study, but more information is needed. The Aurora Study is designed to help researchers learn more about the safety of GTX-102 and the effects it may have on:
GTX-102 is not currently approved by any regulatory authority for use in treating AS and may only be used in approved clinical research studies such as this one.
For more information, please visit clinicaltrials.gov

(up to about 6 weeks)
(about 11 months) GTX-102
“Open-label” means that both the participant and the study doctor will know what treatment is being given to them. There is no placebo (a substance that looks like the investigational treatment but does not contain any medication) or sham control (a procedure that mimics how the investigational treatment is administered but does not actually administer it) in this study, and all participants will be administered the same investigational treatment.
Assessments, testing, and investigational treatment
Discussion about changes in your loved one’s health, other medications/therapies
At study visits, assessments and procedures will be done to check your loved one’s health. This may include but is not limited to:
AS assessments
Neurological examinations

Physical exams

Vital sign measurements
Blood, urine, and cerebrospinal fluid sample collections

Electrocardiograms (heart activity measurement)
Electroencephalograms (EEG brain activity measurement)
Questionnaires

Assessments for adverse events (side effects)
Clinical Study for Children and Teenagers Living with Angelman Syndrome (AS)
Evaluating an investigational therapy called GTX-102 as a potential treatment.To be eligible for study participation, individuals with AS will need to meet the following requirements:

4 to 17 years of age
Confirmed diagnosis of mutation-type AS
If the individual has been previously treated with an ASO or gene therapy, they will not be eligible to participate in this study. Please discuss any previous treatments with the study team to see if your child may be able to participate. There are some additional requirements, which the study team will explain to you.
GTX-102 has been evaluated in a prior clinical study, but more information is needed. The Aurora Study is designed to help researchers learn more about the safety of GTX-102 and the effects it may have on:
GTX-102 is not currently approved by any regulatory authority for use in treating AS and may only be used in approved clinical research studies such as this one.
For more information, please visit clinicaltrials.gov

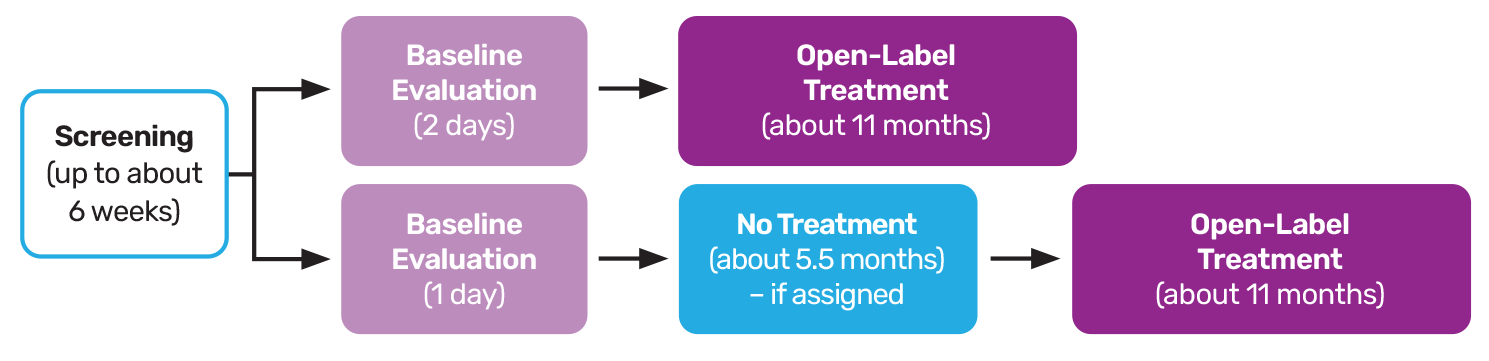
“Open-label” means that both the participant and the study doctor will know what treatment is being given to them. There is no placebo (a substance that looks like the investigational treatment but does not contain any medication) or sham control (a procedure that mimics how the investigational treatment is administered but does not actually administer it) in this study, and all participants will be administered the same investigational treatment.
Assessments, testing, and investigational treatment
Discussion about changes in your child’s health, other medications/therapies
At study visits, assessments and procedures will be done to check your child’s health. This may include but is not limited to:
AS assessments
Neurological examinations
Physical exams
Vital sign measurements
Blood, urine, and cerebrospinal fluid sample collections
Electrocardiograms (heart activity measurement)
Electroencephalograms (EEG brain activity measurement)
Questionnaires
Assessments for adverse events (side effects)
To learn about current and future studies, you only need to fill out the form once.
*All fields are required.
By submitting this form, you confirm that a diagnosis of AS has been made by a medical professional.
If you would like to know more about your privacy and the data you submit and data we collect while you are on this website, please see below or visit our privacy policy.
Our Patient Enrollment Liaison (PEL) team will contact you by email or phone within one business day to discuss your study eligibility and answer questions.
If you choose to be contacted by email, be sure to check your spam folder for an email from [email protected], if it’s not in your regular inbox.
Email our PEL team about current and future medical research opportunities.


Patient portrayal.
Angelman Syndrome (AS) is a rare genetic neurodevelopmental disorder that is caused by a lack of expression of the maternal copy of the UBE3A gene on chromosome 15q11-13 in the brain. Children with AS have intellectual disability, severe speech impairment, and seizures throughout their lives.
If you are thinking about joining a study, our Patient Enrollment Liaison (PEL) team is here to help. Every member of our PEL team has a robust professional background and clinical expertise and experience to help answer your questions. Your PEL team member will assist in exploring Ultragenyx clinical and non-interventional clinical trials and related sites where you could be considered for enrollment. Your PEL is here to make the process as easy as possible.
To speak with a PEL about current and future medical research opportunities, please email our PEL team at [email protected].
When you voluntarily submit your personal data, Ultragenyx stores and processes this information in an electronic database consistent with our Privacy Policy. Ultragenyx will use this information to contact you to fulfill your request regarding Ultragenyx research opportunities.
Clinical and nonclinical studies provide invaluable information to treat, diagnose, or discover new breakthroughs in treating rare and ultra-rare diseases. By choosing to participate in research, you may help advance treatments, provide crucial insights, and ultimately improve the quality of life for others living with a rare or ultra-rare disease. You might also benefit from learning more about your condition, gain access to specific care, or achieve faster access to treatment. All efforts of participation, big and small, can make a difference.
Ultragenyx is leading the future of rare disease medicine. We leverage our experience, insight, and commitment to help move the rare disease community forward. Our primary goal is to provide medicines to those with limited options, and to help patients face rare disease head on with courage and confidence.
Ultragenyx is not responsible for the content or privacy policies of third-party websites outside of its control. Content from Ultragenyx Patient Advocacy (ultrarareadvocacy.com) is intended for a U.S. patient and caregiver audience. Content from the Ultragenyx corporate website (ultragenyx.com) is intended for investors and the general public within the U.S. The information provided on these Ultragenyx sites may not apply to your country. Please contact your physician to learn more about these topics. Thank you for visiting our Clinical Trial site. We hope your visit was informative and enjoyable.
Do you wish to continue?